
My Curriculum Vitae
Thank you so much for coming to my page.
If you want to keep in touch with me, I hope that you will send me
e-mail (yuzy.matsuo@gmail.com).
I am working as research fellow at Cell
Regulatoin Lab in Cancer
Research UK London Research Institute.
I will struggle and enjoy my next science in UK.
Personal
and contact information
Name:Yuzy Matsuo
Birthday:17/2/1983 (around 30 age. orz. www)
Place of birth:South
Shimabara city,
Nagasaki, Japan
Hobby: Fishing, Mountaineering, Walking & Jogging, Reading books
and Doing scientific
experiments.
A motto:Keep on runnning, keep on fighting , where there`s a will,
there`s
a way.
My favaorite proverb:
You can accomplish anything if you'll do it. Nothing will be
accomplished unless you do it.
If something was not accomplished, that's because you didn't do
it.
(成せばなる、成さねばならぬ何事も、成らぬは人の成さぬなりけり(上杉鷹山))
人は城、人は石垣、人は堀。情けは味方、仇は敵なり(武田信玄)。
Education
and Professional Experimence
2005 B.S(Biochemistry) Fuculty of Agriculture,
Saga
University, Saga, Japan
2007 M.A(Yeast Genetics & Molecular biology) Fuculty of
Africulture, Saga University,
Saga, Japan
2008-2010 JSPS research fellow (DC2)
2007-2011 Ph.D.student (Molecular and cellular biology) , The United
Graduate School of Agricultural Science, Tottori University, Tottori,
Japan
2011- Guest researcher (Ph.D (Agriculture)), Department of Molecular
and
Functional
Genomics, Centre of Integrated Research in Science, Shimane University,
Shimane, Japan
Committee
Service,etc.
1.Professional Societies
Japan Society for Bioscience, Biotechnology, and Agrochemistry (JSBBA).
The Molecular Biology Society of Japan
Yeast Genetics Society of Japan
2.Honors and Awards
2008-2010. Research Fellowship for Young Scientists , Japanese Society
for the Promotion of Science
2010. Tyugoku-Sikoku Student encouragement award, Japan Society for
Bioscience, Biotechnology, and Agrochemistry.
Funding Information
Completed
2005-2006 Genetic analysis of nuclear protein quality control system in
fission yeast.
The JAPAN SCIENCE SOCIETY, Sasagawa research grant.
Role:PI (Mentor=Dr.Satoshi Katayama in Saga University)
The major goal of this study is to identify several genes contributed
to nuclear protein quality control
in fission yeast.
2008-2010 Molecular biological analysis of nuclear protein quality
control system in fission yeast.
JSPS, Grant-in-Aid for Young Scientists.
Role:PI (Mentor=Dr.Makoto Kawamukai in Shimane University)
This principle objective of this study is to analyze the molecular
mechanism that play a pivotal role in
nuclear protein quality control in fission yeast.
Invited Presentations
2010. Invited speaker, 27th tyugoku-sikoku area conference of Japan
Society for Bioscience, Biotechnology, and Agrochemistry.Hiroshima,
Japan
Bibliography
Original paper
1. Yuzy Matsuo,Kazuhide
Asakawa,Takashi Toda,and Satoshi Katayama.
A
Rapid Method for Protein
Extraction from Fission Yeast.
Biosci.Biotechnol.Biochemistry, Vol70, p1992-1994, 2006.
This protocol is fortunately cited in several papers. It was
especially glad for me to have been cited by Sir
Paul Nurse's current article. Furthermore, the almost similar
protocol is
published from Dr.M.Yoshida Lab.
In particular, this method is very powerful when you construct tagging
strains. Because glass-beads and proteinase inhibitor cocktail are not
neccesarry to prepare the protein extract in this protocol, we can
handle a lot of samples at the same time (it is especially useful for
checking tagging strains). In addition, it is possible for us to
extract
total protein directly from S.pombe cells grown in not only liquid
medium but also on solid medium (YES, YPD, EMM, SC etc).
2. Yuzy Matsuo, Hayafumi
Kishimoto, Tomitaka Horiuhci, Katsuhiro Tanae and
Makoto Kawamukai.
Simple
and Effective
Gap-Repair Cloning Using Short Tracts of Flanking Homology in Fission
Yeast.
Biosci.Biotechnol.Biochemistry, Vol74, p685-689, 2010.
Unfortunately, nobody except for me have been cited this article.
However, this method is very powerful when you perform the one-step
cloning of multiple DNA fragments to construct a fusion gene. The
principle and procedure of Gap-Repair Cloning is detailed in Dr.Y.Nagano
and Dr.H.Moriya
homepage.
Furthermore, Dr.Moriya Lab showed that GRC
is simple and effective for plasmid construction in fission yeast.
3. Yuzy Matsuo, Hayafumi
Kishimoto, Katsuhiro Tanae, Kenji Kitamura, Satoshi Katayama and Makoto
Kawamukai.
Nuclear
Protein Quality is Regulated by the Ubiqutin-Proteasome System
through the Activity of Ubc4 and San1
in Fission Yeast. The Journal of
Biological Chemistry, Vol.286, p13775-13790, 2011.
This study have been my main project since I was a master-course
student in Saga university ,2006.
This present work shows that the existence of a nuclear protein quality
control systems mediated by the ubiquitin-proteasome system in fission
yeast Schizosaccharomyces pombe
for the first time.
Although protein
quality control (PQC) systems have been extensively studied in the
cytoplasm(e.g. ERAD pathway and CHIP pathway etc.), nuclear PQC
system are not well understood.
In the study
of nuclear PQC system, Dr.Gardner
et.al achieved the pioneering work;
At first, they identified that San1 is
the ubiquitin ligase (E3) contributed to the nuclear PQC in budding
yeast. Next, they recently showed that San1 directly recognizes and
targets the misfolded proteins for proteolysis using conformational
plsticity of disorder (Disorder
targets misorder in Nuclear PQC). Furthermore, current several papers showed
that San1 contribute to PQC not only in the nucleus , but also in the
cytosol in budding yeast.
In addition, Dr.Iwata et.al suggest that UHRF-2 is an
essential E3 ligase for the nuclear pQ degradation as a component of
nuclear
PQC machinery in mammalian cells. All data from their (Drs.Gardner and
Iwata) and our study
indicate that the nuclear PQC via the ubiquitin-proteasome system is
widely conserved from yeast to human. Although there are a lot of
enigma in the study of nuclear PQC system, I believe firmly that the
study will be more
interesting and exiting field. If I have the
oppotunity(time,grant,infla) , I want to struggle to solve the problems.
Fortunately, my paper publised in JBC has been cited by the recent Dr.Gardner's
paper. This very exciting paper demonstrate that budding yeast
nuclear PQC
ubiquitin ligase San1 recognizes exposed hydrophobicity in its
substrate.
4. Katsuhiro Tanae, Tomitaka Horiuchi, Yuzy
Matsuo, Satoshi Katayama, and Makoto Kawamukai
Histone
chaperone Asf1 plays an essential role in maintaining genomic
stability in fission yeast.
PLoS ONE.
5. I am writing down the new paper
related to the latest our study.
Presentations
(from 2006)
- 6th international fission yeast meeting pombe2011, Boston,
Massachusetts, USA,2011.(selected oral talk and poster)
- Job seminar(Postdoc interview), Cancer Research UK, London
research institute, UK, 2011. (oral talk)
- British yeast group meeting 2011, Brighton, UK, 2011.(poster)
- Biochemistry&Molecular Biology(BMB)2010, Kobe, Japan,
2010.(poster)
- 43th Yeast genetic forum(YGF)2010, Nara, Japan, 2010.(oral talk)
- 27th Meeting of JSBBA in tyugoku-sikoku area , Hirosima, Japan,
2010.(invited talk)
- BMB2009, Yokohama, Japan, 2009.(poster)
- 42th Yeast genetic forum 2009, Tsukuba, Japan, 2009.(oral talk)
- 5th international fission yeast meeting pombe2009, Tokyo, Japan,
2009.(poster)
- BMB2008, Kobe, Japan, 2008.(oral talk&poster)
- BMB2007, Yokohama, Japan, 2007.(poster)
- 2007 Meeting of JSBBA, Tokyo, Japan, 2007.(selected oral talk)
- 4th international fission yeast meeting
pombe2007, Copenhargen, Denmark, 2007.(poster)
- 24th Yeast Work Shop, Kagawa, Japan, 2006.(poster)
- 47th Meeting of the Japanese biochemical society in
tyugoku-sikoku area, Matsue, Japan, 2006.(talk)
- 2006 Meeting of JSBBA, Kyoto, Japan,2006. (talk)
How to extract
total protein from S.pombe by
NaOH extraction method
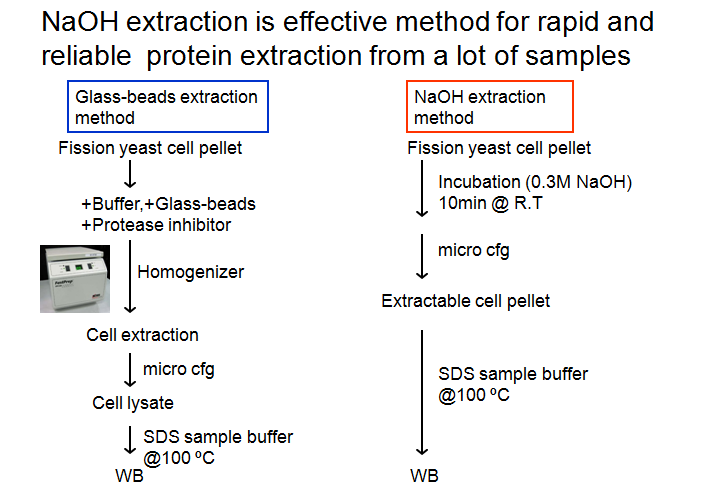
1. 分裂酵母をYES液体培地などで液体培養する or YESプレートなどにストリークする。
2. 約1.0×10の8乗相当の分裂酵母細胞を集める(micro
cfgの場合@RT,7,000rpm,1min)。→なるべくフレッシュな細胞がよい。
3. 滅菌水1mlで洗浄してから、再び集菌する。
4. 滅菌水0.3mlを加えて細胞をよく懸濁してから0.6M NaOHを0.3ml加えてよく混合する。
5. 室温で10min静置培養する。
6. micro cfg(@RT,10,000rpm,2min)後、上清を除去してから70μlのSpecial SDS
buffer(組成は上記論文を参照)を加える。
7.
まずチップの先端でペレットをかき混ぜてバラバラにしてから、静かにpipettingで細胞を懸濁する。→アルカリ処理後の細胞は非常に溶解にしくいの
で注意。
8. @100℃で3~5minボイル後、-20℃保存。SDS-PAGEやWestern blottingに供する量は10μlほどでよい。
How
to perform gap-repair
cloning
(GRC) in S.pombe
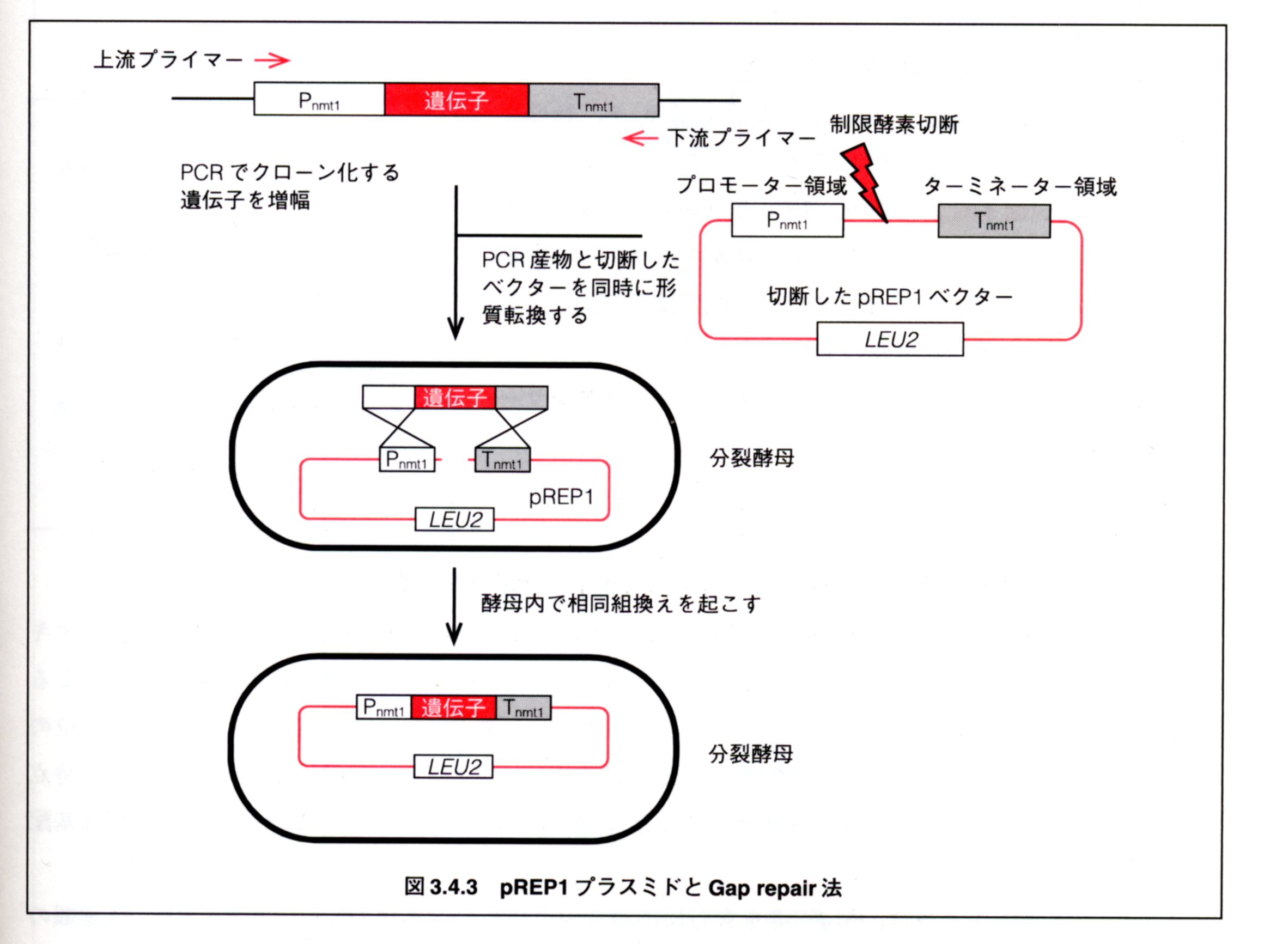
上記図は「ポ
イントがわかる分子生物学第2版(Drs.川向誠 & 真野佳博 編著)」より転載。
GRCの原理は詳細は上記のHPに分かり易く、詳しく説明されているので説明は最小限にとどめ、
ここでは概念図と手順の概略をまず述べる(上図3.4.3)。
(1)
PCRでクローン化したい遺伝子(プロモーター、タグなど)に挿入したいベクター部位の相同配列を付加して増幅する。
(2) 制限酵素あるいはInverse
PCRでベクターの挿入したい位置にgapが入った線状DNAを調製する。
(3) PCR産物と線状ベクターを同時に分裂酵母に形質転換する。
(4) 酵母からプラスミド回収して大腸菌にレスキューする。
(5)
大腸菌よりプラスミドを回収して、制限酵素処理やシーケンスでインサートの構造確認する。
GRC
の実験手順/操作の流れ

Fishing
Blog 「Fish on』
開高健先生には遠く及ばないが、一釣師として敬意をもって
タイトルを真似させていただく。釣りキチ達の山陰の釣りの記録である。

Memory of Mountaineering 「Because
it is there」
ジョージ・マロリーには遠く及ばないが、登山好きの一人として
山陰地方での登山を記録する。

Acknowledgements
Thank
you very much for my
masters,
mentors, family, friends and collaborators
Owining to their helps and encouragements, I was able to
publish several
papers and can get a Ph.D without giving up and going
soft.
佐賀から数えて5年くらいやっているメインの仕事の
Full papaerの論文を
やっとアクセプトさせることができた。少し肩の荷が下りたというかホッとしている。
自分が不甲斐ないばかりに論文執筆にてまどり、研究をもっと深く掘り下げられなかったことは
残念であるが、生涯忘れられない一報であることは間違いない。
川向先生を始め、佐賀大学時代にお世話になった沢山の先生方、JSPS felloeshipや家族などへ
の感謝はしてもしつくせない。本当にありがとうございました。これからもご指導とご鞭撻
のほど宜しく願います。
自分の努力、意思、行動による部分もあるが、多くの方々の協力と尽力あっての一報だと
痛感した。また、チームリーダーとして小さいながらもラボとは独立のグラントを頂いて
一つのベクトルにむけてドライブしていく経験ができたことはPIを目指す自分にとって貴重なもの
だったと思う。
博士進学後、様々なトラブルがあり途中精神的に腐れかけ挫折しかけれども、何とか腐らずに最後まで諦めずに
アクセプトまで貫き通したことに自信を持ち、もっともっとoriginality
の高い優れた研究を遂行するために自らに試練を与えてこれからも向上と精進していく所存である。
精神的タフネスとpoorな生活に対する耐性はかなりあがったのでハングリー精神にさらに磨きをかけて貪欲
に上昇と向上して更なる高みを目指したい。








